PROMES-CNRS researchers have further advanced their lithium-ion battery recycling using concentrated solar heat. The team has now shown they can recover 76% of the lithium and up to 99.3% of the cobalt at 1100°C.
Working with the French battery recycling company SNAM, the PROMES team has modeled and has now demonstrated a proof of concept for recycling lithium-ion batteries using concentrated solar pyrometallurgy for carbothermal reduction, which they presented at SolarPACES 2025
“Our cobalt recovery rate of 99.3% is comparable to or better than what is typically achieved with conventional fossil-fuel-based pyrometallurgy,” lead researcher Ahmed Benamar confirmed.
The lithium recovery at 76% is also much better than the recovery rate in the conventional process – which is just 30%.
“This demonstrates that our solar-assisted process can match or exceed traditional methods while offering significant environmental benefits through reduced CO₂ emissions.”
This battery recycling concept originated at PROMES-CNRS
The field is only a few years old. In 2023, PROMES lead researcher Gilles Flamant first demonstrated that CST-driven carbothermal reduction for battery recycling could work. At the time, he was just beginning to consider how using a temperature sequence might affect the process.
“Perhaps it is possible to use various temperatures, or at one high temperature in a single step?” Flamant mused in an interview with SolarPACES.
Indeed, gradually increasing the temperatures, designed to allow different reactions to occur sequentially at each step, was behind the success of the current work.
“In our current research, we are implementing a precisely controlled temperature sequence (900°C, 1000°C, and 1100°C) to optimize the reduction process,” said Benamar.
“Temperature control is a critical aspect of our pyrometallurgical process. We use a PID controller to manage the temperature by adjusting shutter blades, ensuring precise control over both heating and cooling rates. This level of control is essential for achieving consistent and reproducible results.
The original work in 2023 did not isolate and deeply study the carbothermal reduction step in isolation, or rigorously characterize what was happening to individual elements at different temperatures. But it established the fundamental principles of concentrated solar for battery recycling using pyrometallurgy. This work now builds on that original concept, using a more refined experimental approach that includes detailed process optimization and validation.
Why solar for carbothermal pyrometallurgy?
Today’s battery recycling relies on fossil fuels (either burner or fossil fuel-based electricity) to supply the high temperatures required for pyrometallurgy, which is not sustainable in the long term.
Pyrometallurgy involves smelting, roasting, and calcination to separate valuable metals from impurities, playing a crucial role in the recycling of iron, steel, and batteries. But when heat is supplied by fossil fuels, it is energy-intensive and emits greenhouse gases. Concentrated solar now can reach these temperatures over 1000°C.
Chemical dissolution (hydrometallurgy) can recover metals (particularly Cobalt and Lithium) more selectively, but it generates hazardous liquid wastes.
There is a need for a clean, scalable recycling method for the volumes the world will soon produce — the lithium-ion battery market is projected to nearly triple from $54 billion to $140 billion between 2024 and 2033.
Both metals, cobalt and lithium, are scarce and increasingly valuable
The Democratic Republic of Congo holds 98% of the world’s cobalt reserves, and mining it is inherently brutal work in small, unpredictable artisanal mines that cannot be systematically mined with machinery. The dust causes lung diseases.
Lithium mining is less environmentally problematic, but it’s also geopolitically concentrated among very few nations.
How the demo worked
Carbothermal reduction in pyrometallurgy uses a carbon source – graphite in this case (but biomass char can also be used) – as the reducing agent to reduce metal oxides into elemental metals. Graphite is a strong reducing agent; it has a strong chemical affinity for oxygen and will pull it away from the metal oxides when given enough thermal energy to drive the reaction.
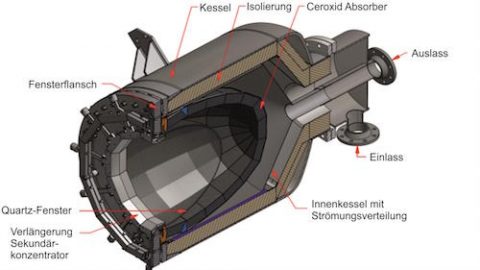
Concentrated solar energy supplied the heat directly to over 1000°C to perform the carbothermal reduction in an oxygen-free container.
For this on-sun proof-of-concept test, the team used a 25-square-meter tracking mirror that followed the sun, redirecting its radiation onto a 2-meter parabolic reflector, to refocus the energy to up to 1100°C at the focal point, where the sample was enclosed in a sealed, transparent reactor. The temperature was precisely controlled by adjusting the shutters to regulate the amount of refocused sunlight reaching the sample.
Lithium-cobalt oxide and graphite, mixed in a 1:1 molar ratio, were placed in small alumina crucibles and heated at a rate of 50°C per minute to a sequence of targeted temperatures of 900°C, 1000°C, and 1100°C, and held there for 45 minutes, then cooled.
This sequence of temperatures causes the carbon to ultimately strip the metal oxides of their oxygen, which disperses as a gas, leaving the metals.
While cobalt was successfully recovered as a solid metal with 99.3% recovery rate, lithium proved trickier: its recovery peaked at around 70% by 900°C and actually declined as the temperature rose, apparently because lithium species vaporize as a gas at extreme temperatures, something their thermodynamic modeling hadn’t predicted.
The researchers are working on finding a fix for this partial lithium escape.
“Our simulations indicated that lithium vaporizes as both atomic lithium gas (Li) and lithium oxide gas (Li₂O),” Benamar specified in a follow-up email.
“During cooling, these gases react with CO₂ or CO in the atmosphere, reforming as Li₂CO₃ (the white powder observed). This understanding has helped us refine our approach to lithium recovery.”
The goal now is to understand the fundamental behavior well enough to refine the process.
Another unexpected challenge they already solved was that the alumina crucible containing the sample unexpectedly combined with the lithium.
“To fix the contamination issue with alumina crucibles, we switched to using MgO crucibles, which successfully eliminated the contamination problem. This change has allowed us to achieve cleaner separation of elements and more consistent recovery yields,” he said.
Next up: solar pyrometallurgy for EV batteries
Nickel Manganese Cobalt (NMC) batteries are used in most modern electric vehicles. The team is now working on a recycling technology for this increasingly commercially prevalent battery chemistry and on the development of a pilot-scale solar reactor.
“We are still gathering data, but our preliminary results are promising,” Benamar noted. “With the projected accumulation of end-of-life NMC batteries in the next five to 10 years, developing effective recycling methods for this chemistry is becoming urgent.”
Flamant foresees that commercialization would see many midsize or smaller solar battery recycling plants, rather than a few large ones, greatly reducing the capital expense for concentrated solar startups. And the timing is perfect.
“You have a kind of delay between the development of battery uses and the production of waste,” he said. “We are now just starting to see an increase in the quantity of waste batteries that must be treated. The quantity isn’t really big right now, but in five or 10 years, it will be. Just in time for a solar process.”
These small solar pyrometallurgy battery recycling plants would be suitable for many regions around the world, those with at least 1800 kWh/per square meter annual DNI. All of Southern Europe, India, Africa, much of China, and most of Australia, the Southwest US and South America could generate these temperatures with concentrated solar for these recycling plants.