In the future, the heat from highly concentrated solar energy could be used for a solar pyrolysis technology to recycle lithium-ion batteries
As a new industry, lithium-ion battery recycling with pyrolysis could be heated by concentrated solar energy from the start
One of the hurdles for concentrated solar thermal energy to decarbonize heat-based industrial processes is that most of these industries, from steel and cement making to chemicals and food processing, have been around for a hundred years or more, and fossil-fueled heat grew up with them over that century, and so fossil heat became the entrenched technology.
But lithium-ion battery recycling using heat (pyro-metallurgy) presents an opportunity for concentrated solar heat to get in on the ground floor of a new industrial process and compete with fossil-fueled heat while both are at a more similar, relatively nascent stage. Pyro-metallurgy is one of the two ways batteries are being recycled. (The other uses chemicals, hydro-metallurgy)
Research at PROMES-CNRS laboratory, led by Gilles Flamant, has just confirmed the feasibility of a solar-driven pyro-metallurgical process to recycle the lithium, nickel, cobalt, and other valuable components from lithium-ion batteries. His teams at PROMES do groundbreaking work in industrial decarbonization with very high-temperature solar heat.
“My first interest in lithium-ion batteries was because I think we will have a large quantity of material that must be treated in the next decades,” said Flamant in a call from France.
“And I was just thinking in a general way of how solar thermal technology can play a big role in the decarbonization of industry. In this particular domain, the treatment of wastes and by-products and, more generally, the field of solar recycling of materials, is one of my directions of research now. In industry, hydrometallurgy is often selected instead of pyrometallurgy since this last process is a very energy-intensive route. Using concentrating solar thermal energy can be a game-changer in this field.”
The team successfully used high-temperature solar heat to produce a metal alloy containing lithium, cobalt, nickel, and manganese (iron). A full description of the test results is in a new conference paper from PROMES, A Solar Pyro-Metallurgical Process for Li-ion Batteries Recycling. Proof of Concept.
Lithium-ion batteries have four main components: anode, cathode (current collectors), separator, electrolyte, and contain strategic metals like lithium, cobalt, and nickel at the cathode.

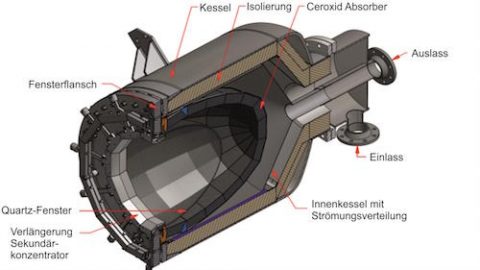
The 1.5 kW solar reaction chamber at PROMES where the proof of concept test for solar-heated pyrolysis for lithium battery recycling was carried out Image©A Solar Pyro-Metallurgical Process for Li-ion Batteries Recycling. Proof of Concept.
How the experiment worked
The team conducted the proof of concept solar pyrolysis test using the 1.5 kW solar furnace at PROMES. The study successfully demonstrated for the first time a pyrometallurgical process for recycling strategic metals from lithium-ion batteries using the heat-based form of solar energy. Generally, in these kinds of solar thermal processes, sunlight is first reflected off many mirrors (heliostats) in a solar field to a solar receiver, and the heat is transferred to a reactor where the reaction takes place. (The difference between pyrolysis and combustion)
It was a three-step process:
1. Thermal pre-treatment by solar pyrolysis at 580°C for 30 minutes to remove binders, organic impurities, and separate electrode materials and collector foils (copper and aluminium).
2. Mechanical separation of cathode and anode materials to extract metallic foils and the pyrolysis product from the cathode, leaving a “black mass.”
3. Carbothermal reduction at 1000°C for 30 minutes which smelts the black mass with graphite from the anode, producing a Ni-based alloy.
The two heated steps, 1 and 3, were conducted in sequence in the same chamber under argon gas. For this initial test, the team used the small 1.5kW reactor available at PROMES. It is a beam-down system, one heliostat reflects sunlight up to a parabola to further concentrate the solar radiation onto a reaction chamber underneath.
“We used beam-down here at PROMES because we have small furnaces in which we can make some proof of concept at low power and low cost,” he said. “But it is not limited to that. It could also be horizontal like a receiver in a tower.”
What they learned and what’s next
Flamant’s team found that the temperature will need to be maintained within a specific narrow range. For instance, in the first step, when they pyrolyzed the cathode material at below 580°C, the separation of the metallic foil and the black mass is not possible. And at 600°C, the metal collector became brittle. So the recommended pyrolysis temperature will have to be kept at between 580°C and 590°C.
“And we must also be sure that the temperature is the same in the whole volume of the crucible in order to have a product with homogeneous composition,” Flamant explained.
They achieved this even overall temperature by adjusting the position of shutter blades controlling the incoming solar heat reflected by the heliostats into the crucible where pressure and atmosphere composition are controlled and measured.
As the much-cited PROMES-CNRS Research Director, Flamant has led many teams doing high-temperature solar thermochemical research over the years. He modeled the kind of open-ended “divergent” creativity it takes to solve complex engineering challenges in discussing one of the next steps; separating out the component metals.
“Perhaps it is possible to use various temperatures – or at one high temperature in a single step?” he mused.
“Or is it mixed? Possibly… I don’t know. The proof of concept is a first attempt to actually make it with solar energy. Next, we will need to separate lithium from the cobalt and nickel. But, what is the quantity we can treat in one set, and also, is it possible to make it in a continuous way? It is one of the numerous open questions … Perhaps it is possible only in batch processes, that is to say, replacing crucible by crucible.”
Other challenges future teams will need to solve include refining the temperature for the smelting step, engineering the continuous process, investigating the reaction mechanisms, and preventing pollutants from escaping.
“Even if you don’t put oxygen in the system, you need to extract the gas, which is the product of the decomposition during the pyrolysis step,” Flamant explained.
“It’s not very difficult, but we have to manage this type of emission to be sure that we don’t create dangerous compounds emitted in that first low-temperature step, and that is why we have to check what is the composition of this gas and if that composition is modified with heating temperature and heating rate.”
Small-scale battery recycling in the sun-drenched South

Flamant suggests that small concentrated solar pyrolysis plants for lithium battery recycling heated by a solar field of heliostats would be dispersed throughout Southern Europe, in Spain, Portugal, Italy, Greece and the South of France, where PROMES itself is located. Image©DLR
A nearby small battery recycling company was impressed that no contaminants at all were produced in the metal alloy, even in this first result. That firm is now helping and providing waste batteries to further this solar pyrolysis battery recycling research.
Flamant foresees many midsize or smaller solar battery recycling plants rather than a few large ones as gigafactories producing batteries in Europe. Given the solar resource best for concentrated solar technologies of at least 1800 kWh/per square meter annual DNI, he sees European solar battery recycling dispersed throughout Southern Europe, in Spain, Portugal, Italy, Greece and the South of France, where PROMES itself is located.
As a very new industry, the need to recycle batteries will grow at the same time as their research is refined.
“You have a kind of delay between the development of batteries uses and the production of waste. We are now just starting to have to increase the quantity of waste batteries that must be treated. The quantity is not really big up to now, but in five or 10 years, it will be. Just in time for a solar process,” he said.
Fraunhofer Chile to develop a solar concentration system to recycle aluminum