Abstract:
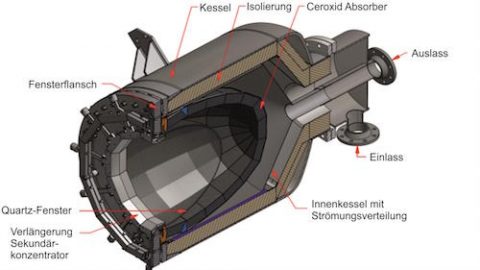
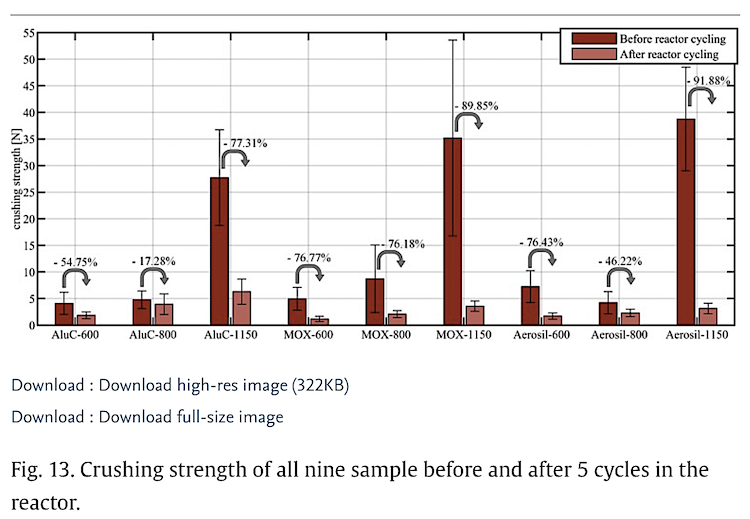
Thermochemical energy storage systems have a high potential to support a future energy supply based on renewables by providing comparably high storage capacities and option for long-term, cold and loss free storage, that allows a flexible utilization in several application areas. Due to its cost efficiency and abundance the reaction system Ca(OH)2/CaO is currently intensively researched. However, the handling of the material in its typically powdery form exhibits some major challenges under cyclic hydrothermal loading due to its high cohesivity and strong tendency towards agglomeration. To increase the bulk performance during cycling, various approaches to stabilize the particles are being investigated, most of them involve the addition of high fractions of thermochemically inert additives and thus high losses in energy density. This paper investigates the approach of coating pre-granulated Ca(OH)2 with nanostructured inorganic oxides by a low-cost mixing process followed by thermal treatment, which requires a low mass fraction of additives compared to the total material. The samples are comparatively analysed regarding their cycling stability, morphology as well as regarding their bulk performance and mechanical stability in a fixed bed reactor. It was found that the shells formed from coating materials increase the stability of the granules and, due to their permeability to the reactants, do not hinder the reaction. Especially the sample coated with nanostructured Al2O3 and thermally post-treated at 600 °C, has the greatest potential to reconcile cycling stability with structural integrity and mechanical stability. A constant conversion rate of around 88% could be measured over several cycles, which is also comparatively high. A constant conversion rate implies maintenance of structural integrity, which could also be confirmed by SEM micrographs.
Jashari, A., Afflerbach, S., Afflerbach, K., & Krumm, W. (2023). Structural stabilization of granular Ca(OH)2 by coating with nanostructured additives for thermochemical cycling in a fixed reaction bed. Energy Conversion and Management: X, 18, 100367. https://doi.org/10.1016/j.ecmx.2023.100367
Read the full paper at Energy Conversion and Management