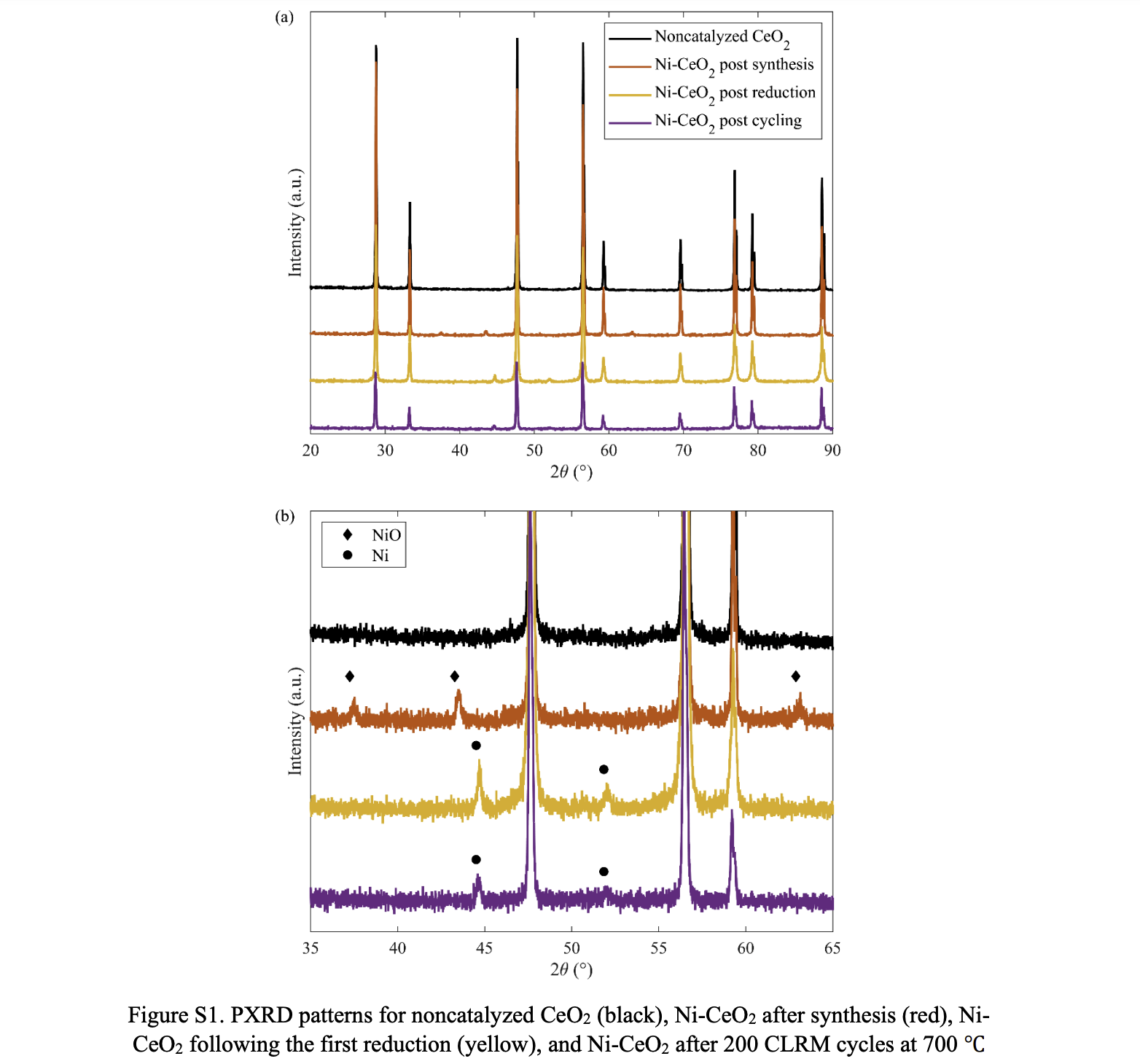
Abstract:
Leveraging solar thermal energy to drive the chemical looping reforming of methane (CLRM) is a promising method of efficiently and selectively reforming methane to produce syngas using renewable energy. In this work, the role of catalytically active nickel in reaction kinetics, conversion, selectivity, and total syngas production during CLRM over Ni–CeO2 is investigated. Through thermogravimetric analysis (TGA), metallic nickel is shown to help enhance partial oxidation of methane (POM) reaction rates as a result of a lower activation energy reaction mechanism at all oxygen nonstoichiometries, compared to CeO2. For example, reduction rates of Ni–CeO2 at 700 °C are comparable to CeO2 at 900 °C, and no reaction is observed for CeO2 at 700 °C. Further, extended cycling with Ni–CeO2 demonstrated stable reaction rates and yields during CLRM at 700 °C, and SCO remained above 0.98 for the duration of experimentation. Utilizing a larger-scale packed-bed reactor system, Ni–CeO2 also demonstrated comparable methane conversion, syngas production and selectivity to CeO2, but at notably lower operating temperatures, i.e., T ≤ 800 °C. Higher rates of coking were observed during POM over Ni–CeO2; however, all carbon was removed in the subsequent step and accumulation was not observed during extended cycling. A parametric study of gas velocity, temperature, and inlet partial pressure of methane is also presented to examine the effect these operating conditions have on conversion, selectivity, and syngas production. Notably, a tradeoff between conversion and the quantity of syngas produced was observed as gas velocity increased; however, time response of conversion indicates an ideal reaction cut-off time exists where high rates of syngas production can be achieved simultaneously with near complete methane conversion.